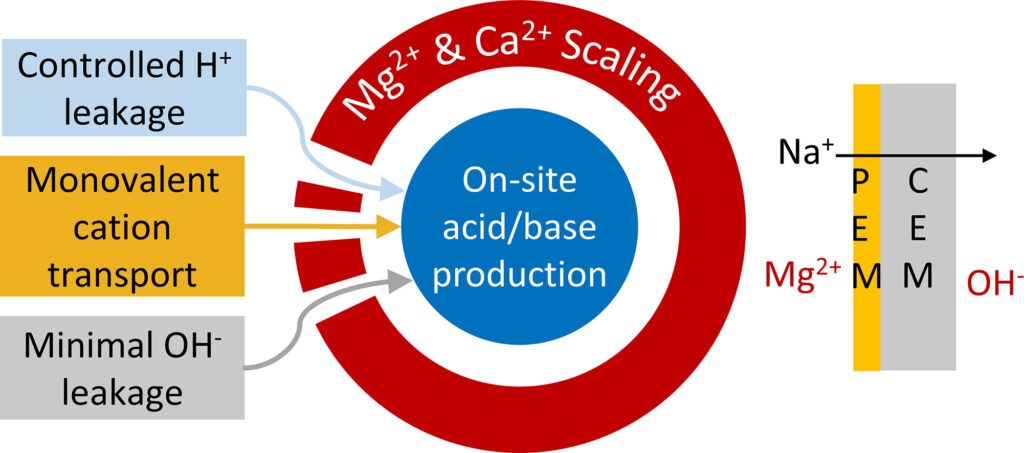
The ability to produce acids and bases on-site at low energy and cost is of great interest to seawater reverse osmosis desalination plants. Acid is needed to lower the seawater pH to mitigate scaling in reverse osmosis (RO) modules. Base is needed to shift the boron equilibrium in the permeate of the first RO stage towards borate, which can then be rejected by the membrane in a second RO stage. We investigated the direct use of synthetic seawater in a bipolar membrane electrodialysis (BMED) process to produce acids and bases on-site. When synthetic seawater was used in a conventional BMED stack, Mg(OH)2 scaling occurred inside the BMED stack, specifically in the salt compartments and the cation-exchange membranes (CEMs). The scaling resulted in a decline in current efficiency and raised the energy consumption of the BMED stack compared to using a pure NaCl stream. To mitigate the scaling, we used CEMs coated with a PAH/PSS multilayer to limit the transport of multivalent cations from the salt to the base compartment. Moreover, the salt compartment was acidified with a fraction of the produced protons to prevent the precipitation of Mg2+ and Ca2+. In the first run, the modified BMED stack successfully produced a 0.2 M OH− and a 0.2 M H+ solution at 85% current efficiency and 0.05 kWh/mol OH−. However, repeated operation showed a gradual decline in performance due to OH− leakage through the CEM. To ensure long-term process stability, CEMs with improved OH− retention must be developed for the BMED application. Read more here: https://doi.org/10.1016/j.desal.2025.119788